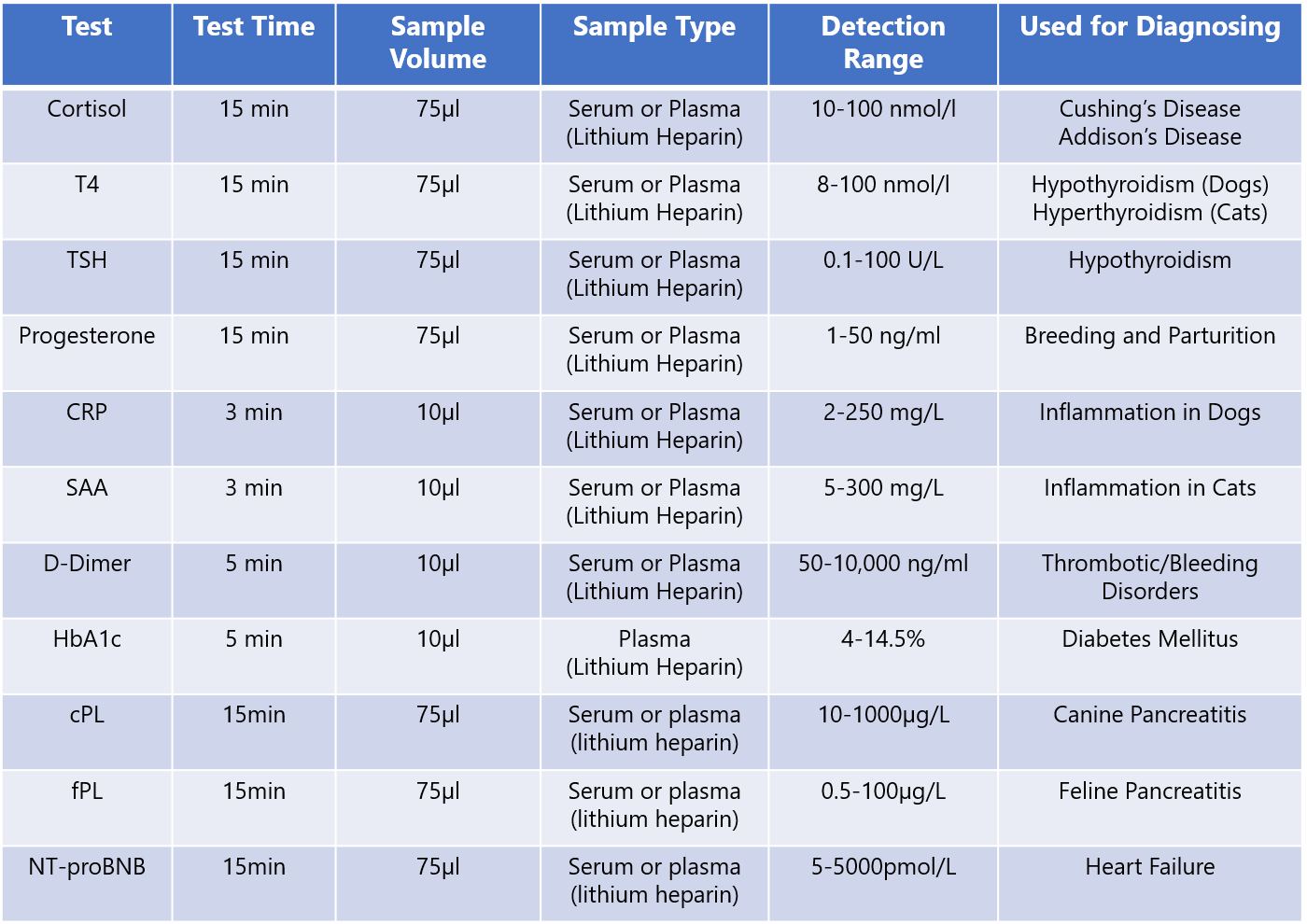
T4 (10 test) for Insight V-IA Analyse
InSight V-IA Thyroxine(T4)Rapid Quantitative Test er en fluorescens immunoassay til som anvendes til InSight V-IA Veterinær Immunoassay Analyzer for kvantitativ bestemmelse af T4 koncentration i serum eller plasma. Testen anvendes som hjælp til at vurdere bugspytkirtlens funktion.
Description
Anvendelse
InSight V-IA Thyroxine(T4)Rapid Quantitative Test er en fluorescens immunoassay til som anvendes til InSight V-IA Veterinær Immunoassay Analyzer for kvantitativ bestemmelse af T4 koncentration i serum eller plasma. Testen anvendes som hjælp til at vurdere bugspytkirtlens funktion.
Test principper
Mængden af T4 læses af InSight V-IA Veterinary Immunoassay Analyzer, hvorefter T4 -koncentrationen udtrykkes i nmol/L eller µg/dl.
Opbevaring og stabilitet
- Test sættet opbevares ved 4-30 ℃ frem til udløbsdatoen.
- Når posen er åbnet, skal testen udføres indenfor 1 time.
- Hvis testen opbevares i køleskab, skal der gå 30 minutter før anvendelse, så testen kan opnå stuetemperatur.
Prøvesamling og forberedelse
Testen kan udføres med enten serum eller plasma.
1. Efter venepunktur (flebotomi), opsamles en hel blodprøve i et blodopsamlingsrør. Hvis der anvendes en plasmaprøve, skal du bruge et blodopsamlingsrør indeholdende lithium heparin antikoagulant. Hvis der anvendes en serumprøve, skal du bruge et serum-separationsrør.
2. Separer serum eller plasma fra blodet indenfor 2 timer efter at blodprøven er taget. Hvis en prøve er meget hæmolyseret, anbefales det at tage en ny prøve.
3. Testen skal udføres umiddelbart efter prøvetagningen. Hvis testen ikke kan udføres inden for 2 timer efter blodopsamling, skal testen opbevares ved 2 til 8 ℃, og maksimalt i 48 timer. Hvis prøverne skal opbevares i længere tid, så skal de opbevares ved en temperatur på under -20 ℃.
Når en test skal anvendes efter langtidsopbevaring og/eller nedfrysning, så skal den optøs til stuetemperatur og herefter blandes godt inden den anvendes. Bemærk at prøven ikke må nedfryses igen efter optøning.
Test procedure
Se brugervejledningen til InSight V-IA Veterinary Immunoassay Analyzer for komplet vejledning til analysatoren.
- Sæt testkassetten på en ren, vandret overflade.
- Sørg for, at testpatronens partinummer stemmer overens med ID-chip nummeret og indsæt herefter ID Chippen i analysatoren. Vær opmærksom på ikke at røre ved spidsen af ID Chippen. Tryk på ‘Læs ID-kort’ på testskærmen.
-
Pipetter 75 μl af prøven over i bufferrøret og bland forsigtigt. Kraftig omrøring og skumdannelse bør undgås.
-
Pipetter 75 μl blandet prøvefortynding over i prøvebrønden i testpatronen. Undgå at der dannes bobler i prøven.
-
Se afsnit V i InSight V-IA veterinær immunassay Analyzer brugervejledning for detaljer.
a) Quick Test Mode: Indstil timeren til 15 minutter, og start timeren med det samme efter tilsætning af prøveblandingen til prøvebrønden. Når timeren er færdig, indsættes testpatronen i patronholderen på analysatoren. Klik herefter på ‘Test’. Analysatoren scanner testenheden automatisk og viser herefter testresultatet.b) Standard Test Mode: Indsæt testenheden i patronholderen på analysatoren straks efter tilføjelse af prøven til prøvebrønden, skal du klikke på ‘Test’. Analysatoren begynder at nedtælle og læser automatisk testresultatet.
-
Resultatet vises på hoved skærmen og udskrives automatisk.
Fortolkning af testresultater
InSight V-IA Veterinary Immunoassay Analyzer beregner test resultatet automatisk og viser koncentrationen af T4 på skærmen.
Yderlige oplysninger findes i brugervejledningen til InSight V-IA Veterinary Immunoassay Analyser.
Reference område:
| nmol/L | ug/dL | ||
| Hund | Lav Normal Høj |
< 15 15 – 50 > 50 |
< 1,2 1,2 – 3,9 > 3,9 |
| Kat | Lav Normal Høj |
< 15 15 – 60 > 60 |
< 1,2 1,2 – 4,7 > 4,7 |
OBS: Hver klinik bør oprette et reference område, som er repræsentativt for de dyr som testes på klinikken.
Kvalitetskontrol
Hver InSight V-IA T4 Rapid Quantitative Test indeholder en kvalitetskontrol, som automatisk udføres hver gang en prøve testes.
Hvis kvalitetskontrollen registrere et ugyldigt resultat, vil analysatoren vise en fejl meddelelse og prøven skal tages om.